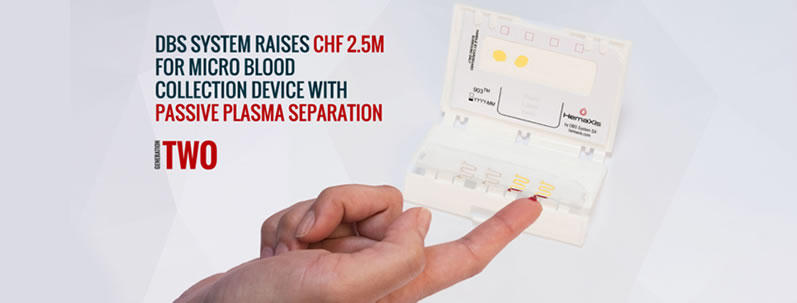
DBS System raises CHF 2.5M to produce revolutionary micro blood collection device capable of true passive plasma separation
DBS System announced it closed its Series A at CHF 2.5M which included the direct investment of a Swiss pension fund. DBS System produces blood collection devices that leverage advanced microfluidic technology, greatly simplifying and reducing the cost of blood sampling without modifying existing lab analysis workflows. With this investment, DBS System will industrialize its second generation HemaXis™ blood collection device that passively separates plasma or serum from whole blood thus dramatically increasing the range of tests capable of being run on dried blood spot samples by certified laboratories.
The CHF 2.5M investment round was completed with CHF 1.0M coming from DBS System’s existing shareholders and private business angels and the additional CHF 1.5M was led by investiere.ch with CHF 0.8M coming from investiere.ch’s business angels and CHF 0.7M from the Swiss pension fund Nest. Nest is building a Swiss venture portfolio in partnership with investiere.ch and is systematically co-investing alongside investiere.ch’s business angels. Nest’s investment in DBS System is the first example of this partnership.
DBS System aims to establish itself as the leading manufacturer of medical devices for collecting and preparing micro samples of whole blood, plasma and serum with its patented HemaXis™ platform of microfluidic devices. DBS System is already producing and selling its first-generation device, HemaXis DB for whole blood collection and is establishing partnerships with major labs in Europe and the US.
Building on the advantages of their first-generation device, HemaXis DP will be the world’s first microfluidic device capable of producing ultra pure plasma or serum at the fingertip with a truly passive method that eliminates the need for centrifugation, filtration or any other assistance. DBS System’s patented technology promises to vastly expand the potential of micro blood samples and unleash the power of labs to test and monitor your health.
“The advantages of our technology are clear: the device can be used by anyone in a non-medical environment, blood samples are reliable, secured and standardized, logistics are simplified, and it fits within the existing workflow, all together resulting in significant cost savings for labs” says CEO Eric Ödman. “This model is beneficial for us and our partners because they can expand their market by reaching more patients and it enables us to leverage them to build awareness and prescribe the use of our devices through their distribution networks in an estimated addressable market size of USD 20 Billion”.