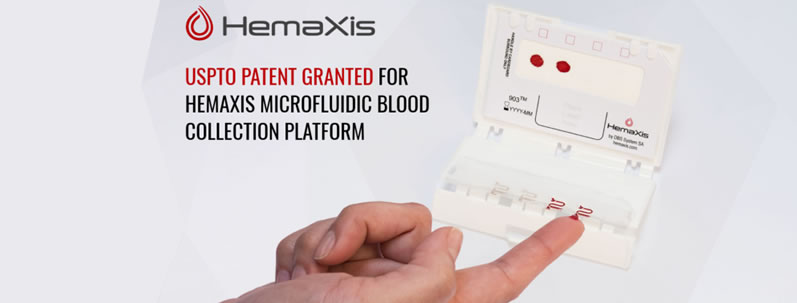
USPTO Patent Granted For HemaXis Microfluidic Blood Collection Platform
We are proud to announce the USPTO patent covering our 1st Generation micro blood collection device, HemaXis DB, has been fully granted under “Device and Method for Dried Fluid Spot Analysis”. This further secures the market potential for our innovative HemaXis™ platform and the opportunity to leverage its microfluidic based technology. See HemaXis DB in action below and then register with us under the Connect menu option to order a free evaluation kit.